Training structure
Faculty of Science
Presentation
Program
Thermodynamics and phase equilibria
2 creditsOPTION 1
4 creditsChoose 2 out of 3
Supplements in solution chemistry
2 creditsCrystallography I
2 creditsAnalysis of biomolecules by mass spectrometry
2 credits
Polymers
2 creditsAdvanced inorganic materials
2 creditsSolutions, colloids, interfaces
2 creditsLiquid NMR spectroscopy and X-ray diffraction
2 creditsChemometrics, statistical data analysis, experimental design
2 creditsOrganometallic chemistry and heteroelement chemistry
2 creditsMethodology for characterizing materials
2 creditsCoordination chemistry and organic chemistry
2 creditsProfessional projects – project monitoring
8 credits
2-4 month M1 internship with thesis defense/report in English
10 creditsOPTION 2
4 creditsChoose 2 out of 4
Fundamentals of Process Engineering
2 creditsBio-based chemistry
2 creditsLiquid-liquid extraction: kinetics and thermodynamics
2 creditsMedicinal chemistry
2 credits
Innovative synthesis and extraction processes
2 creditsThermal and mechanical properties of materials
3 credits20hIntroduction to modeling
2 creditsNanomaterials
2 creditsCommunication and professional integration
2 creditsElectronic and optical properties
2 creditsHybrid and structured materials
2 creditsDistributed systems
2 credits
Thermodynamics and phase equilibria
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
- Review of thermodynamics of single-component systems.
- Basic concepts of thermodynamics in multicomponent systems. Chemical potential, Gibbs-Duhem relation, variance.
- Concepts related to thermal analysis techniques used to construct binary/ternary diagrams: ATG, ATD, and DSC
- Construction and interpretation of binary phase diagrams based on thermodynamic quantities. Gibbs free enthalpy, pressure, and temperature diagrams as a function of the composition of the binary mixture. Liquid-liquid, liquid-vapor, and solid-liquid mixtures.
- Phase transformations: first- and second-order transitions, critical points. Examples.
- The supercritical state: definition, thermodynamic properties, most widespread industrial applications.
- Construction and interpretation of ternary phase diagrams: variance, definitions of ternary eutectic, first and second order peritectic, isothermal section, study of alloy cooling.
Hourly volumes:
CM: 13
TD: 7
Supplements in solution chemistry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This course on solution chemistry aims to introduce the various concepts necessary for studying complex liquid mixtures used in separation chemistry. The approach taken is mainly thermodynamic. In particular, we explain the role of concentration effects, beyond the ideal laws that apply only to dilute solutions.
CM: 12 H
Tutorial: 8 hours
Crystallography I
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science

This lecture, delivered entirely in English, provides a basic introduction to crystallography and electron diffraction for beginners. X-ray diffraction is an important characterization technique in modern chemistry; the majority of crystalline structures in inorganic and organic solids have been solved using this method. It is therefore important for all students to understand its basic concepts and instrumentation. The course provides explanations and principles of X-ray diffraction together with the geometry and symmetry of X-ray patterns. In addition to the interaction principles of X-rays and matter, it covers how to obtain quantitative intensities for single crystal and powder diffraction patterns. It naturally includes an understanding of lattice planes and the reciprocal lattice concept together with the Ewald sphere construction. Furthermore, it provides a basic understanding of the Fourier transform relationship between the crystalline structure and the diffracted intensities, as well as the reciprocal lattice concept.
Electron diffraction is a complementary technique to X-rays that provides information in terms of symmetry and geometry on the materials studied. In this course, we will therefore approach the description of the method for obtaining electron diffraction patterns and their interpretation. We will be able to obtain the lattice parameters, the reflection conditions, as well as the groups of possible spaces.
This lecture also serves as the introductory part to the lecture Electron Microscopy and Crystallography II.
CM: 14
TD: 6
Analysis of biomolecules by mass spectrometry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
Knowledge of the latest mass spectrometry techniques for the qualitative analysis of organic molecules and biomolecules.
1) Description of fundamental principles (Ion science and technology):
- Ionization techniques
- Analysis techniques
- Tandem mass spectrometry (MS/MS)
- LC/MS and LC/MS/MS couplings
2) Application in the context of biomolecule analysis and monitoring of organic chemistry reactions.
Hourly volumes*:
CM: 3 p.m.
Tutorial: 5 hours
Polymers
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
Polymers are all around us: we eat them, we wear them, and we use them to construct extremely complex buildings. From mature technologies to the most innovative materials, polymers are a crucial building block for constructing the world of tomorrow. In this course, we will cover several aspects such as the controlled synthesis of polymers and cross-linked materials, surface modification using polymers, some characterization tools suitable for polymers, and finally a last section developing the latest advances involving polymers.
Hourly volumes:
CM: 1:00 p.m.
Tutorial: 7 hours
Advanced inorganic materials
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The HAC720C module covers "advanced inorganic materials" in five main sections. Thefirst section is devoted to general information on inorganic materials and discusses structure-property relationships, with particular attention paid to chemical bonding, real crystals, and polycrystalline solids. The different classes of inorganic materials are described. Thesecond part focuses on ceramic materials (definitions and properties) and their synthesis (raw materials including clays, shaping, drying and debinding, sintering); a distinction is made between traditional ceramics and technical ceramics (synthesis methods for oxide and non-oxide ceramics). Thethird part covers glass (classification and synthesis methods) and glass-ceramics (devitrification and soft chemistry); their properties and applications are also discussed. Thefourth part is dedicated to metals: properties of metals and metal alloys; metal nanoparticles; and catalytic materials. Part5 is devoted to inorganic materials developed for energy; ceramics (oxides and non-oxides; nanostructured) and metal hydrides are described (properties and synthesis) through several examples and in the context of their applications (accumulators, hydrogen storage, and carbon dioxide capture).
Hourly volumes:
CM: 1:00 p.m.
Tutorial: 7 hours
Solutions, colloids, interfaces
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This course unit enables students to acquire basic knowledge and cross-disciplinary skills in the field of colloids and interfaces, which are common to the various tracks of the Master's degree in Chemistry (Materials Chemistry, Separative Chemistry, Materials and Processes, ICAP Cosmetics Engineering, Biomolecular Chemistry). It is also offered to international students enrolled in the SFRI program at the University of Montpellier, where courses are taught in English. An introductory presentation on basic notions and concepts will enable students to discover and better understand the main physicochemical properties of colloidal dispersions, associative colloids, and macromolecular solutions, as well as the parameters and phenomena governing stability in colloidal dispersions and mixed solution-colloid systems. This will be followed by interdisciplinary practical teaching based on the flipped classroom principle to help students build and deepen their knowledge through individual and collective analysis of the various applications of colloidal and interfacial phenomena and systems.
Hourly volumes:
CM: 7
TD: 13
Liquid NMR spectroscopy and X-ray diffraction
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
NMR:
Liquid-phase NMR (Nuclear Magnetic Resonance) is an essential spectroscopic analysis method for chemists, enabling them to determine the structure of small organic molecules or macromolecules in solution, study dynamic phenomena, and more. The aim of this course unit is to understand the phenomena involved in this technique and to relate them to the various structural information accessible by this method. The goal is to be able to use the spectral data from this analysis to elucidate the structure and stereochemistry of organic molecules or polymer structures, or to monitor reactions.
X-ray diffraction:
X-ray diffraction is a powerful, non-destructive technique for characterizing the crystalline structure of materials. It can also provide crystallographic and structural information such as lattice parameters and atomic positions. This includes all crystallized materials such as ceramics, materials for energy and information storage and conversion, as well as organic molecules and metal complexes (interatomic distances and angles, stereochemistry (chirality, stereoisomerism, etc.), intra- and intermolecular bonds, etc.). The objective of this course unit is to provide an introduction to crystallography and diffraction, with the aim of understanding the operation and characteristics of an X-ray diffractometer, as well as interpreting diffraction patterns (structural analysis, lattice parameters).
Hourly volumes:
CM: 10
TD: 10
Chemometrics, statistical data analysis, experimental design
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This course will cover the fundamental concepts and practical tools related to chemometrics through: - statistical data analysis;
- the laws of probability;
- confidence interval estimation;
- parametric and nonparametric tests.
An introduction to design of experiments will be offered at the end of the module.
Hourly volumes:
CM: 7 a.m.
TD: 1:00 PM
Organometallic chemistry and heteroelement chemistry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The first part of the course presents the fundamental knowledge of organometallic chemistry of transition metals. It begins with a description of the metal-carbon bond, enabling an understanding of its stability and chemical reactivity. Next, we will demonstrate the power of this synthesis tool for forming C-H, C-C, and other bonds. Examples of their applications in different fields will help students learn about these reactions and their fields of application: fine chemistry, catalytic transformations of industrial importance, synthesis of natural products, and preparation of materials.
The second part of this course is devoted to the chemistry of heteroelements, focusing on silicon, tin, and boron. This part aims to present the different methods of preparing boron-, tin-, and silicon-based reagents, as well as the main transformations carried out with these compounds, with applications in organic synthesis and materials synthesis.
CM: 1:00 PM
Tutorial: 7 hours
Methodology for characterizing materials
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The program of this EU focuses on describing the principles and applications of the main methods for the structural characterization of solids, thin films, surfaces, and interfaces, as well as several examples of applications in materials chemistry. It includes the following techniques.
- Introduction to solid-state NMR (NMR signal, interactions in solid-state NMR, magic angle spinning, NMR sequences, cross polarization, instrumentation, etc.)
- Electron microscopy: principles and applications of scanning and transmission electron microscopy and related techniques (EDS microanalysis).
- Spectroscopic methods: Raman spectroscopy, photoelectron spectroscopy, X-ray spectroscopy (XAS, XRF, etc.), Mössbauer spectrometry.
Hourly volumes:
CM: 10 a.m.
Tutorial: 10 a.m.
Coordination chemistry and organic chemistry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This teaching unit is dedicated to deepening the foundations of organic chemistry and coordination chemistry covered in L3 and to acquiring concepts related to molecular engineering and molecular chemistry. The teaching unit consists of lectures and tutorials. Students will prepare for certain lectures and tutorials using course materials provided, enabling them to participate fully in the lectures and tutorials, understand the concepts presented, and acquire the necessary skills. The progression program and activities will be proposed. For students who have not studied the basics of coordination chemistry and organic chemistry, documents will be made available.
Coordination chemistry: The course will cover various aspects of transition metal and lanthanide complexes, molecular materials (polynuclear complexes and coordination polymers with extended structures (MOFs, etc.)) as well as their properties and applications. Structural aspects, bond descriptions, properties, and aspects related to stability and reactivity will be addressed. Emphasis will be placed on the complexation effect and the stability of metal, lanthanide, and actinide complexes with certain ligands for applications in the biomedical field (imaging and therapy), decontamination (nuclear field), etc. The electronic (relaxivity, magnetism) and optical (absorption, luminescence) properties of these complexes will be discussed and placed in the context of applications in various fields, such as imaging, electronics, sensors, etc.
Organic Chemistry: The course builds on the knowledge acquired in the Bachelor's degree and will use a reasoned study approach to address the main reaction mechanisms in organic chemistry, providing a common foundation for all students in the Master's in Chemistry program. The main processes (substitution, addition, elimination, transposition, etc.) and their essential characteristics and applications to mechanistic sequences will be examined. This course should provide students with general tools for analyzing mechanisms (ionic, radical, concerted) in order to understand these mechanisms in all their variety.
Hourly volumes:
CM: 1:00 PM
Tutorial: 7 hours
Professional projects – project monitoring
Level of education
Bachelor's degree
ECTS
8 credits
Training structure
Faculty of Science
The professional project bridges the gap between traditional practical work and internships in laboratories or companies. It takes the form of a supervised project consisting of placing students in a professional situation through collaborative (group) work based on carrying out a project in response to a problem set by a company, local authority, association, or academic. It is part of the core curriculum of the Master's in Chemistry and is carried out under the supervision of a member of the teaching team (academic or industrial). Conducted throughout the semester, this project aims to connect and consolidate the knowledge and skills acquired during the Bachelor's and early Master's programs through this professional situation. These scenarios will be directly related to the Master's program chosen by the students. In addition to chemistry-specific skills, other interpersonal, organizational, and communication skills intrinsically linked to project management will also be acquired, equipping students for their future professional lives.
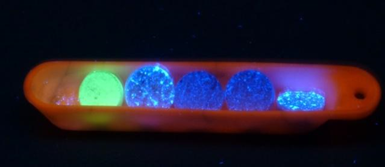
Addressing a research issue: example of a summary of new phosphorescent materials.
Hourly volumes:
CM: 5 hours
Tutorial: 5 hours
Practical work: 40 hours
2-4 month M1 internship with thesis defense/report in English
Level of education
Bachelor's degree
ECTS
10 credits
Training structure
Faculty of Science
The internship in semester 8 of the Master 1 in Biomolecular Chemistry aims to familiarize students with careers in life sciences research. Students will have the opportunity to complete this introductory research internship in academic or private laboratories. Subject to prior approval by the teaching team (internship topic related to the Master's program and appropriate environment/resources), students may seek a host team in an academic setting at one of the institutes of the Chemistry Cluster at the University of Montpellier (IBMM, ICGM, , etc.), in academic laboratories outside the University of Montpellier (in France or abroad), or in the private sector (chemical, pharmaceutical, and agri-food industries, biotechnology laboratories, etc.).
Fieldwork: 2 to 4 months of internship
Fundamentals of Process Engineering
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The goal of this course is to enable students with a background in chemistry to understand the fundamentals of process engineering.
The course consists of two main parts that are illustrated by the same process.
In the first part of the course, a drying process will be used to introduce the most common heat and mass transfer phenomena found in process engineering, from which the dimensionless numbers can be derived. In the second part, the thermodynamic properties of the air/water vapor mixtures will be used to derive basic dimensioning rules for the same drying process.
This course will be taught entirely in English.
Hourly volumes:
CM: 10
TD: 10
Bio-based chemistry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The following topics will be covered:
- Bio-based solvents
- Fuels derived from biomass
- Antioxidants derived from lignin
- Metal catalysts derived from plants
- Surfactants obtained from renewable resources
- Examples of industrial applications of enzymatic synthesis
Hourly volumes*:
CM: 15
TD: 5
Liquid-liquid extraction: kinetics and thermodynamics
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
A general approach to liquid-liquid extraction will be developed through thermodynamics and kinetics concepts with a view to understanding the mechanisms responsible for extraction and the processes taking place at the liquid-liquid interface. The fundamental aspects of other types of extraction (liquid-solid, supercritical fluid, distillation) will also be addressed.
Hourly volumes:
CM: 12 p.m.
Tutorial: 8 hours
Medicinal chemistry
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The aim of teaching medicinal chemistry is to introduce students to the key stages in the process of developing molecules with biological activities. In particular, a description of the interactions involved, the concept of pharmacophores, bioisosterism, etc., as well as structure-activity relationship studies will be covered, enabling students to consider appropriate strategies and structural modifications.
Hourly volumes:
CM: 3 p.m.
Tutorial: 5 hours
Innovative synthesis and extraction processes
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This teaching unit is shared by MI students in the Master's in Chemistry program: ICAP P1, ICAP P2, MAT P1, MAT P2, and BM (semester S2) courses. The following topics will be covered:
- The 12 Principles of Green Chemistry and units of measurement in Green Chemistry;
- Synthesis strategies in sustainable chemistry;
- Alternative or eco-friendly solvents for synthesis and extraction;
- Unconventional activation techniques and applications.
CM: 13
Tutorial: 7 hours
Thermal and mechanical properties of materials
Level of education
Bachelor's degree
ECTS
3 credits
Training structure
Faculty of Science
Hours per week
20h
The mechanical and thermal properties of materials are central to many applications in the field of energy materials. After an introduction to these different fields of application, this course unit aims to define the various concepts necessary for understanding both the mechanical and thermal properties of materials, limiting itself to bulk materials.
Hourly volumes:
CM: 11 a.m.
TD: 9 a.m.
Introduction to modeling
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
General overview of the most commonly used calculation and modeling methods in the field of solid-state chemistry according to the spatial and temporal scales that can be studied with them:
(1) Quantum calculations (Hartree Fock, Post-Hartree Fock methods, DFT),
(2) Force field-based modeling (atomistic and coarse-grained),
(3) Hybrid QMMM and AACG modeling.
Presentation of different calculation techniques: static and optimization calculations, molecular dynamics, and Monte Carlo.
The EU will offer lectures and practical classes. Two practical modeling assignments will be offered: modeling techniques in classical mechanics and quantum calculations.
CM: 11 a.m.
TD: 9 a.m.
Nanomaterials
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This teaching unit is dedicated to the presentation of inorganic materials and nanomaterials intended for use in the biomedical field (imaging, therapy, implants). This teaching unit builds on the knowledge acquired in teaching unit HAC930C (Development of Materials for Health). It aims to develop health issues and inorganic materials and nanomaterials in diagnosis, therapy, and well-being. Strategies for developing the inorganic materials and nanomaterials of the future based on theranostics and multifunctionality, as well as smart materials, will also be addressed.
The EU includes lectures and tutorials. Students will be offered a group project on the (theoretical) study of inorganic materials or nanomaterials for health.
CM: 11
TD: 9
Communication and professional integration
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
This EU will address, in small groups or on an individual basis, teaching tools and best practices related to communication and professional integration, through:
- assessment of knowledge, skills, competencies, interpersonal skills, and motivations;
- awareness of job search techniques;
- writing resumes and cover letters;
- rules for oral and written communication;
- job interview simulations.
Scenarios directly related to the sectors of activity targeted by the courses of the students concerned will be offered.
Practical work: 20 hours
Electronic and optical properties
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The electronic and optical properties of solids are central to many applications in the fields of energy (photovoltaic panels, passive coolants, etc.), light production (white diodes, lasers, etc.), and electronics (components, microprocessors, etc.). After an introduction to these different fields of application, this course aims to define the various concepts necessary for mastering both the electronic and optical properties of materials, which are essential for understanding the most modern technologies.
Hourly volumes:
CM: 11 a.m.
TD: 9 a.m.
Hybrid and structured materials
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
Hybrid materials are a new family of materials combining organic ligands that connect inorganic entities, and are increasingly being studied at both a fundamental and applied level.
As part of this course unit, two main categories of hybrid materials will be covered:
- Coordination Networks and Metal-Organic Frameworks
- Organosilicon/carbon materials
CM: 10 a.m.
Tutorial: 10 a.m.
Distributed systems
Level of education
Bachelor's degree
ECTS
2 credits
Training structure
Faculty of Science
The theoretical knowledge necessary for understanding, formulating, and implementing dispersed systems will be detailed in this module. The physicochemical principles governing the preparation and stability of solid-liquid and liquid-liquid dispersions will be detailed in accordance with specifications and expected usage properties. The various concepts covered include powder dispersibility, modification of the solid/liquid interface to control zeta potential and colloidal interactions (extended DLVO), and rheology of dispersed systems in relation to the state of dispersion. Liquid-liquid dispersion: emulsification, Winsor's R ratio, formulation using the HLD method, and formulation maps.
Introduction to synthesis techniques in dispersed media: emulsion synthesis of nanoparticles, latex, microcapsules, etc.
CM: 11
TD: 9
Admission
Registration procedures
Applications can be submitted on the following platforms:
- French and European students: follow the "Mon Master" procedure on the website:https://www.monmaster.gouv.fr/
- International students from outside the EU: follow the "Études en France" procedure:https://pastel.diplomatie.gouv.fr/etudesenfrance/dyn/public/authentification/login.html


